ZSL
Zoological Society of London
One of the questions I am asked most often is how wildlife pathologists cope with the massive diversity of species that they examine.
It is a very good question; my training was almost entirely centred on mammals (particularly farm and companion animals) with a few chickens thrown in for good measure. At ZSL, I have to diagnose diseases in animals from all over the tree of life, looking at everything from spiders to starfish, from snakes to seahorses! With such great variety, it is impossible to be an expert in everything, so I return to the basic principles of pathology (the science of disease) and anatomy, and extrapolate from those species and diseases which are better understood. I will use this opportunity to talk through a single case in detail.
A juvenile emerald tree boa bred at London Zoo
Our case is a juvenile emerald tree boa (Corallus caninus), a non-venomous snake from the rainforests of South America, that had been bred at London Zoo. The snake was the weakest of its clutch, being the last to learn how to strike-feed and had trouble shedding its skin. The snake became depressed and dehydrated and, despite supportive care from the keeping and veterinary staff, the juvenile was found dead with its head submerged in a pond in its enclosure. Had it drowned? Was it suffering from an infectious disease that could affect other animals in the reptile house? These questions are the reason why wildlife pathologists can be so important to maintaining healthy and high-welfare live-animal collections, and so I set about investigating the death.
Although the adults can grow over 2 metres long, and weigh up to 2 kilos, this juvenile weighed just 60 grams (about the same as a gerbil). That said, my smallest post-mortem examination (PME) so far was just 0.14 g (a male black widow spider), so this was positively gigantic by comparison! Nonetheless, this was going to be a fiddly dissection. The emerald tree boa was too small to easily photograph, so included are some pictures from a PME of a larger corn snake to illustrate this process.

Dissecting a snake
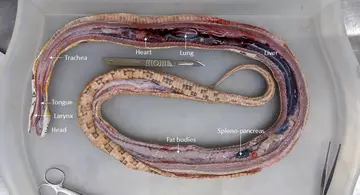
Although their external body plan is unlike ours, snakes perform nearly all the bodily functions that humans do, so you will find almost all of the same organs in them as well. A snake’s organs are only loosely attached to the body wall, meaning after one long incision down the mid-line of the snake, one can remove the internal organs as one big block with the trachea at one end and the cloaca at the other.
All the organs can then be gently dissected apart, allowing for careful examination for evidence of disease (“lesions”).
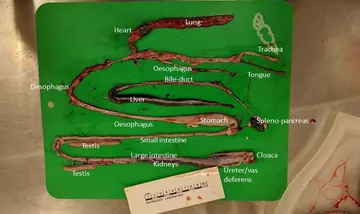
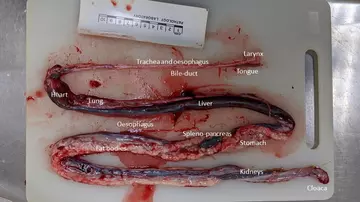
You may recognise the names of many organs shown above, though the shapes are quite different - everything is long and thin! Below is the organ mass of the juvenile emerald tree boa. Everything is much smaller and harder to recognise, but see if you can identify any of the organs. One thing that stands out is that, unlike our corn snake, the fat bodies are almost completely absent in this animal. This indicates that the snake was in poor body condition, which could be caused by chronic disease or may be linked to the animal’s history of difficulty feeding.
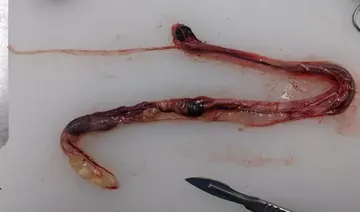
The dissecting process
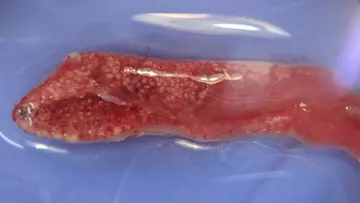
The most significant lesion was found in the cranial (literally “head-end”) sections of the lungs. This lesion was so subtle, that it can only be photographed clearly with a dissecting microscope (Figure 5). Under the dissecting microscope, we can see numerous evenly-spaced cream-white spots covering the lung, creating a honeycomb-like pattern. To understand what these represent, we need a brief aside on the anatomy of snake lungs versus mammalian lungs. Mammals are warm blooded and need to absorb a lot of oxygen in order to feed our metabolic demand. As a result, our lungs are evolved to have a huge internal surface area over which gas-exchange can take place. To achieve this massive surface area, our lungs operate on a fractal, branching system of organisation: the trachea splits into two bronchi, which continue to branch until they terminate in tiny air sacs called alveoli.
Snakes are cold blooded and do not have as high a metabolic requirement as humans, so their lungs are considerably simpler. Instead of branching, the primary bronchus runs the whole length of the lung, with multiple openings at all levels. Meanwhile, the lung takes the form of a sac-like, hollow tube around the bronchus, with a network of low walls forming polygonal “rooms” (known as faveoli) on the inner surface to increase surface area. Picture a honeycomb wrapped around a leaky hosepipe!
Our findings
Now we understand snake lungs, we can see that something white is filling up the faveoli (the honeycomb spots shown above) of the cranial end of the lung. To understand exactly what that white stuff is, we need to use histopathology - the science of using microscopes to examine tissues at very high magnifications. Unlike the dissecting microscope, these microscopes require a complex process of making slides from very thin slices of the tissue and staining them to colour the cells.
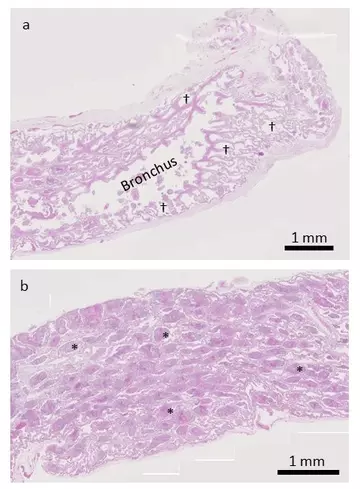
In the less-affected lung (Figure 6a), we can see lots of clear space, representing air in the faveoli. In the worse-affected region (Figure 6b), there is relatively little clear space as the faveoli are filled with red-blue material (the white material seen earlier, only now it is stained to make it easier to see). If we zoom in further, we can see that this material is actually composed of densely packed cells.
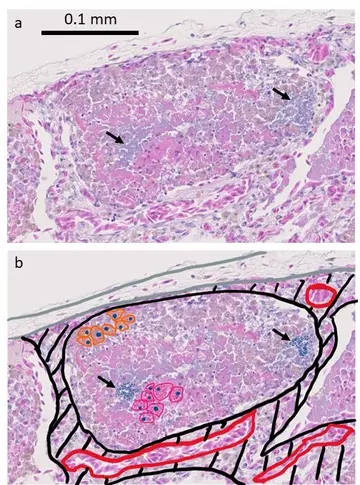
Looking at the histology, the diagnosis becomes clear; bacteria have entered the faveoli and started to damage the faveolar walls. The immune system has responded by sending two types of white blood cell to fight the infection, heterophils and macrophages. These are cells of the innate immune system (they are non-specific and can respond quickly to a range of infections), and “eat” the much smaller bacteria to destroy them. There is no evidence of lymphocytes, the major cell of the adaptive immune response, meaning that this must be an acute (recent) infection. Because the bacteria must have entered the faveoli through the bronchus (i.e. by inhalation) this type of inflammation of the lung is called bronchopneumonia. This must be differentiated from infections that primarily affect the faveolar walls (interstitial pneumonia) as that can be caused by several epidemic viruses that we would definitely not want within our reptile house.
The only question that remains is how the bacteria entered the lungs in the first place, as the respiratory system is lined with defences that would usually prevent this. Once again, the microscope can give us the answer.
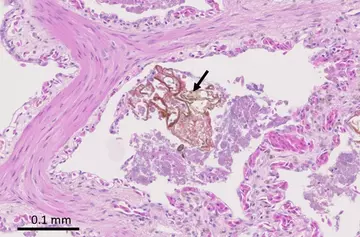
Within some of the faveoli we can find fragments of foreign material (i.e. material not produced within the body) surrounded by bacteria. This suggests that the snake has inhaled food or tank content (soil, plants etc.) that it may have intended to eat. These fragments of material are far bigger than the bacteria that the respiratory defences are designed to filter, and so they carry the bacteria down into the lungs. This process of choking on food or foreign matter is called “aspiration”, so the full diagnosis and cause of death for this animal would be acute aspiration bronchopneumonia. We cannot be certain of the reason for the aspiration, but we do know that the animal had always had problems feeding. However, we can reassure the reptile house that there is no evidence of any underlying viral infections or other undetected diseases that could pose a risk to other snakes in the collection.
