Andrew Cunningham
Deputy Director of Science
Andrew Cunningham, Professor of Wildlife Epidemiology, explains the origins of the coronavirus pandemic, and how we can avoid another pandemic caused by 'zoonotic' diseases.
As Professor of Wildlife Epidemiology, my research explores diseases in wild animal populations.
I investigate which infections circulate naturally, and under what conditions they might jump to become diseases in other species - including humans.
The origins of the coronavirus pandemic
In recent weeks, as governments and the media around the world seek to understand the origins of the coronavirus pandemic, the work of our wildlife health team at ZSL has gained significant attention.
In a series of interviews I have explained how Covid-19 began, how it might have been even worse, and what must be done to minimise the risk of something like this ever happening again.

How did COVID start?
One of the tragedies of the situation is that this pandemic was both predictable and preventable.
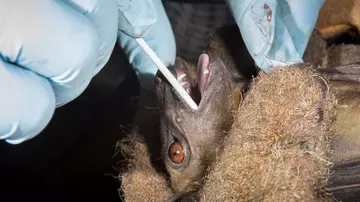
In 2005 I wrote in the British Medical Journal on the dangers of human exploitation of, and closer contact with, wildlife and for the last 15 years I have studied viruses in bats and found them to be a uniquely diverse reservoir of potential zoonoses.
A paper I co-authored in 2013, for example, identified 137 bat viruses of which 61 were known to be capable of infecting people at that time. The phylogenetic data obtained so far indicate that SARS-CoV-2 (the virus that causes COVID-19) is from a species of bat.
The initial progress of the virus is difficult to trace and several other animals – most notably pangolins – have been suggested as intermediary hosts that could have transferred SARS-CoV-2 between bats and humans, although there remains insufficient evidence to support these claims.
And, while authorities have yet to identify patient zero, the human outbreak most likely began at a wildlife market in the city of Wuhan in China.
How can we avoid another COVID?
The highest priority for the short-term protection of human health is to ban these “wet” (live wild animal) markets and to carefully regulate any future legal wildlife trade.
Perhaps the prime example of human overexploitation of wildlife, animals in wet markets are taken from the wild, brought together from different places, transported over large distances and crammed together into cages.
By this stage, they are stressed and immunosuppressed and so excrete whatever pathogens they have in them while also being increasingly vulnerable to infections they might not naturally encounter.
People, who are in intimate contact with the body fluids of slaughtered animals, are also exposed to novel pathogens creating the ideal circumstances for new diseases to emerge.
Now a pandemic, Covid-19 has an estimated case-fatality rate of between 1% and 3% (although the data on this are poor).
This represents a major threat to public health and the scale of the clinical challenge must not be underestimated, but our continued exploitation of the natural world exposes humanity to some much more dangerous diseases.
Ebola, for example, kills around 50% of those infected and Nipah virus around 60%, rising to 100% in some outbreaks.
Both viruses originate from bats.
Solutions to avoid wild animal diseases reaching humans
At the root of the problem is human behaviour, and so changing this provides the solution.
But to know which behaviours to change, we must fundamentally shift our approach to research.
Some of those necessary – like ending the illegal wildlife trade - are large, but others can be tiny.
Covering the areas of shaved tree bark and the containers used to collect date palm sap, for example, stops bats contaminating the sweet liquid drunk by children in Bangladesh and so the risk of Nipah virus spill-over is virtually eliminated.
To find these solutions we need to understand more about wildlife diseases and how humans and wildlife interact.
But we cannot do it alone.
We need support.
We need significant investment in preventative wildlife health research.
With significant investment, we will come to understand more of the risk factors for zoonotic virus spill-over and so - as we can with Nipah - take steps to prevent it happening in the first place without adversely affecting wild animals in which the viruses occur naturally.
75% of all new human diseases identified in recent years began as zoonoses (i.e. originated from animals). We have seen SARS, MERS, Zika, Ebola and now Covid-19 emerge as serious public health threats from wildlife.
How many more warnings do we need?
We are already working to tackle threats to wildlife – and people – around the world. With your support, we can do even more.
Please donate today to help us continue our important work in understanding wildlife diseases and how to avoid similar zoonotic pandemics like COVID-19.